Don’t Let Depression Control Your Life – SPRAVATO Nasal Spray Can Help You Find the Depression Relief You Deserve
When traditional medications aren’t working, a new hope is on the horizon. SPRAVATO (esketamine) is the first and only FDA-approved depression treatment that targets the glutamate system for fast, long-lasting results. Administered as a nasal spray, esketamine is derived from the anesthetic ketamine, which has a long history of being used to treat Major Depressive Disorder (MDD) and treatment-resistant depression.
Because esketamine nasal spray is considered a controlled substance, it must be administered by specially trained medical personnel in a certified clinical setting who can monitor for side effects of SPRAVATO while ensuring patients see the most beneficial results. Most patients receive their esketamine treatment as an outpatient and go home in a couple of hours.
Antidepressants Aren’t Always the Answer

1 in 3
people struggling with depression have Treatment-Resistant Depression (TRD), meaning they don’t respond to medications.

Recovery
with SPRAVATO (Esketamine) nasal spray treatment is 2.5x more likely for patients who failed 2+ medications.
Is esketamine nasal spray an option for you?
Have you had an unsatisfactory response to antidepressants or is a conventional antidepressant not working at all? Are you experiencing unpleasant side effects, or have learned that you cannot tolerate them? If so, esketamine treatment is an alternative form of mental health care that can help you find relief from depression symptoms.
Currently, FDA approval allows for SPRAVATO (Esketamine) to be used together with oral antidepressants, and only for patients who meet specific criteria, which include:
- Those with Treatment-Resistant Depression who have not responded to at least two different oral antidepressants
- Those with major depressive disorder experiencing acute suicidal thoughts
- 18+
Once one or more of these conditions are met, providers can discuss incorporating SPRAVATO treatment into the patient’s customized treatment plan if:
- Their provider believes that SPRAVATO (Esketamine) therapy could work for them and their unique needs for depression or other mental health care.
- The patient is comfortable with trying SPRAVATO nasal spray.
If all the pieces fit, SPRAVATO therapy can be an excellent alternative antidepressant treatment option for depression.
How does SPRAVATO (Esketamine) nasal spray treatment work?
Like antidepressants, SPRAVATO (Esketamine) depression treatment raises the levels of certain chemicals like dopamine, norepinephrine, and serotonin that occur naturally in the brain as neurotransmitters. However, unlike oral antidepressant treatment, SPRAVATO nasal spray increases the brain’s most prevalent chemical messenger: glutamate levels, thereby creating new connections in the brain in order to rapidly improve symptoms. This results in a greater reach and more substantial impact on brain cells with a single dose compared to antidepressants.
SPRAVATO Treatment Patient Results at Mindful Health Solutions
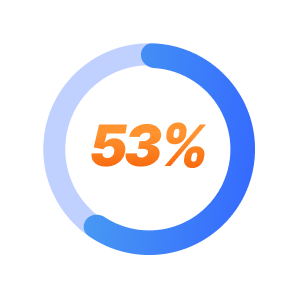
53% of TRD patients experience full remission with SPRAVATO
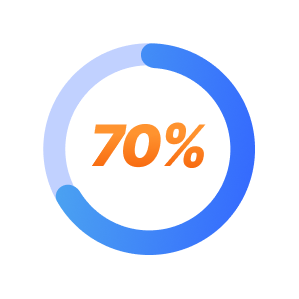
70% of TRD patients respond positively to SPRAVATO
Efficacy & Safety | SPRAVATO®(esketamine) HCP
What Our Patients Are Saying about Esketamine Nasal Spray
“Peacefulness and joy have replaced trauma and depression. [To] People who have traveled this dark journey of depression – there’s light at the end of the tunnel. Just try one more thing. Have hope, one more time.”
Why Mindful Health Solutions for Esketamine Treatment for Treatment-Resistant Depression?
SPRAVATO (Esketamine) nasal spray is a fairly new treatment for depression, and not everyone can provide the same expert and safe medical care as we can at Mindful Health Solutions. As one of the nation’s top SPRAVATO treatment providers for depression care, we have the knowledge and expertise to help you achieve relief and happiness. Our experienced clinical leadership team has been applying innovative treatment protocols through SPRAVATO (Esketamine) nasal spray for years supported by positive outcome data. In fact, Mindful Health Solutions has one of the largest SPRAVATO (Esketamine) patient outcome repositories in the world, with more than 35,000 treatment visits to track our patients’ progress in real time and develop algorithms that help predict the probability of successful interventional treatment outcomes.
Our expert clinical team is committed to helping you find happiness, and will only recommend Spravato (Esketamine) treatment as an option if we believe it will help you finally experience relief. All treatments are administered in a comfortable outpatient setting, ensuring safety and minimal side effects of SPRAVATO. Trust Mindful Health Solutions for safe, effective SPRAVATO therapy on your path to mental wellness – it is our mission to provide innovative and effective care, helping you overcome the challenging symptoms of depression and find joy again.
Frequently Asked Questions
How effective is SPRAVATO therapy?
Wondering, what is SPRAVATO and how fast does it work?
Unlike most conventional antidepressant medications that can take several weeks to show effects, SPRAVATO (Esketamine) nasal spray often begins to alleviate symptoms within a few weeks and sometimes even days. Many patients report a rapid improvement in mood and outlook after treatment.
How is SPRAVATO nasal spray administered?
Each esketamine treatment is administered in our certified SPRAVATO clinics. At the clinic, patients self-administer the nasal spray in a relaxed setting. They are then monitored for side effects of SPRAVATO as well as to ensure optimal results, for at least two hours prior to leaving the clinic.
How do you feel during esketamine therapy for depression?
During treatment, you may feel dissociation, which is a common effect of SPRAVATO (Esketamine). The experience of dissociation can vary from person to person but may include:
- Feeling detached from your body
- Having a distorted perception of time and space
- Experiencing altered perceptions and sensations
- Entering a dream-like state where thoughts may become distorted
- Emotional detachment
Some individuals describe a sense of mental clarity or expanded consciousness.
Is SPRAVATO (Esketamine) covered by insurance?
Yes, because SPRAVATO (Esketamine) for depression is approved by the FOod and Drug Administration (FDA), it is covered by most major insurance providers with prior authorization by your insurance company. Mindful Health Solutions is in-network with Kaiser, Medicare, and most major commercial insurance plans. Contact us to discuss SPRAVATO cost and insurance coverage options.
Eligibility:
- Adults 18+
- Major Depressive Disorder diagnosis
- 2+ antidepressant failures OR intolerable side effects to medication
When will I start to notice results from SPRAVATO (Esketamine) treatment?
Many patients observe a significant improvement after 3 to 8 treatments, with some achieving remission by the 12th esketamine nasal spray treatment. It’s important to keep in mind that each person’s experience with this depression care therapy is unique.
Are there any side effects of SPRAVATO?
The most common side effects of SPRAVATO usually only occur within one to two hours after the nasal spray has been administered. To help ensure the safety of patients, there is a two-hour window of supervision at the clinic following the administration before patients can leave and head home. Esketamine side effects include:
- Sleepiness
- Nausea
- Increased blood pressure
- Bad taste in mouth
- Disassociation
What is the treatment schedule for SPRAVATO (Esketamine) nasal spray?
For Spravato (Esketamine) to become included in your customized treatment plan, our staff will gather information on your symptoms and background, as well as any treatments you have attempted. They will answer any questions you may have and, if you are interested, discuss how the treatment works. From there, you will be able to schedule your treatment sessions.
The FDA-approved treatment plan for SPRAVATO (Esketamine) involves attending sessions twice a week for the first 4 weeks, followed by weekly sessions for the next 4 weeks. We also recommend a tapering protocol and maintenance treatments to help sustain remission. The complete FDA-approved protocol includes 12 treatments, assuming you tolerate the medication well. Your physician will also help you to determine maintenance treatments based on your unique needs.
All treatment plans at Mindful Health Solutions are customized to each patient. Our talented team of clinicians and support staff is dedicated to working with patients collaboratively to find a, esketamine for depression treatment plan that works for them. Our goal is to help patients find relief in a way that feels good.
Is SPRAVATO (Esketamine) used to treat anything other than depression?
While esketamine therapy is only FDA-approved for adult treatment-resistant depression and MDD with acute suicidal ideation, it can also help with other mental health conditions. Many people who get SPRAVATO (Esketamine) for their depression also experience other conditions like OCD, anxiety, or an eating disorder.
Our Chief Medical Officer, Dr. Tobias Marton, explains, “In my experience, it is sort of a ‘rising tide lifts all boats’ kind of thing. As your depression gets better, other aspects of your mental health get a little bit better as well.”
Again, SPRAVATO (Esketamine) is only approved for depression. However, it is possible that you may experience relief in other areas of your mental health when going through treatments.
The FDA-approved treatment plan for SPRAVATO (Esketamine) involves attending sessions twice a week for the first 4 weeks, followed by weekly sessions for the next 4 weeks. We also recommend a tapering protocol and maintenance treatments to help sustain remission. The complete FDA-approved protocol includes 12 treatments, assuming you tolerate the medication well. Your physician will also help you to determine maintenance treatments based on your unique needs.
All treatment plans at Mindful Health Solutions are customized to each patient. Our talented team of clinicians and support staff is dedicated to working with patients collaboratively to find a treatment plan that works for them. Our goal is to help patients find relief in a way that feels good.
For Providers: Can I refer a patient for SPRAVATO (Esketamine)?
Yes! We offer a simple referral process. After you refer your patient to Mindful Health Solutions for esketamine therapy, we'll take care of the rest.
- Assign treatment specialist to provide full support
- Handle insurance authorization and review SPRAVATO price information
- File claims for treatments on behalf of your patient
- Consultation with providing clinician on the treatment process
- Schedule your patients based on their lifestyle needs
- Clinician closely monitors patients throughout treatment
- Collaboration with referring provider and stakeholders
- Patient returned to referring provider for continuation of care
SPRAVATO therapy is only approved for depression. However, it is possible that patients may experience relief in other areas of their mental health when going through treatments.
The FDA-approved plan for SPRAVATO (Esketamine) treatment involves attending sessions twice a week for the first 4 weeks, followed by weekly sessions for the next 4 weeks. We also recommend a tapering protocol and maintenance treatments to help sustain remission. The complete FDA-approved protocol includes 12 treatments, assuming patients tolerate the medication well. We also help to determine maintenance treatments based on their unique needs.
All treatment plans at Mindful Health Solutions are customized to each patient. Our talented team of clinicians and support staff is dedicated to working with you and your patients collaboratively to find a, esketamine treatment plan that works for them. Our goal is to help patients find relief in a way that feels good.
