Transcranial Magnetic Stimulation (TMS)
TMS Therapy: A New Path Forward When Depression Won’t Lift
When you’re struggling with depression, it can be hard to see the light at the end of the tunnel. Sometimes depression keeps its grip even after medications or therapy. TMS offers a different way forward.
TMS gently stimulates the areas of the brain that control mood—helping them work better again—so you can feel more like yourself. There’s no surgery, no anesthesia, and no medication changes required. You stay awake, treatments are quick, and you can get right back to your day. For many people, TMS becomes the turning point: more energy, clearer thinking, brighter days, and the ability to reconnect with life, family, and the things that used to matter.
“I didn’t realize how heavy everything felt—until the weight finally lifted.”
If depression has held you back, if you’ve tried more than two antidepressants without relief, or are experiencing uncomfortable side effects, there is hope—and there are options. TMS may be the next step that helps you move forward.
Give TMS therapy a try and prepare for a new era of health and happiness.
What is TMS Therapy?
TMS stands for “Transcranial Magnetic Stimulation.” This revolutionary depression treatment approach was approved by the Food and Drug Administration (FDA) in 2008 as a safe, non-invasive, non-medication treatment for clinical depression, or major depressive disorder. TMS therapy is designed to help people with mental health conditions who have not found success with medications and/or antidepressants, utilizing magnetic fields to stimulate the specific part of the brain known to control mood.
TMS treatment has extremely limited side effects, especially when compared to traditional medications and antidepressants, so it is also a treatment option for people who may be suffering from side effects while trying to experience relief from their condition.
What does TMS treat?
Since its FDA approval as a depression treatment in 2008, further studies have shown that Transcranial Magnetic Stimulation has the potential to treat a wide range of conditions, many of which can be debilitating. Although the Mindful Health Solutions team focuses primarily on alleviating the symptoms of major depression and treatment-resistant depression, our clinics also provide treatment for other conditions, like:
- Obsessive-Compulsive Disorder (OCD)
- Post-Traumatic Stress Disorder (PTSD)
- Suicide Ideation
There is also evidence TMS treatment may help in recovery from stroke. Research and studies on the impact of TMS therapy for anxiety, OCD, bipolar disorder, and more are continuing, and we at Mindful Health Solutions are excited to be at the forefront of this revolutionary treatment.
How does TMS work?
Individuals experiencing depression are shown to have reduced electrical activity in certain areas of the brain. TMS treatment works by delivering highly targeted, localized electromagnetic pulses via magnetic coils to those areas, including the prefrontal cortex, which is the area of your brain that regulates mood. These electromagnetic pulses stimulate the affected neurons, which in turn releases neurotransmitters and hormones such as serotonin and dopamine. These neurotransmitters work to relieve symptoms of depression. Once these neurons are stimulated by TMS, they continue to release these neurotransmitters for a longer period of time compared to other treatments like antidepressants.

Patients who failed 2 antidepressants are 2x more likely to achieve remission with TMS.
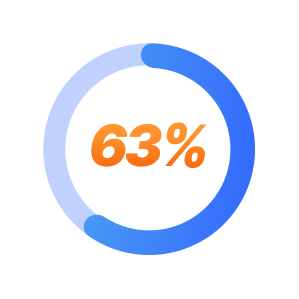
TMS is effective in approximately 63% of patients.

Patients who receive TMS versus conventional medication are more likely to recover from depression at their 12 month follow-up.

What does Transcranial Magnetic Stimulation treatment look like?
TMS is a non-invasive procedure. During a TMS treatment, the patient sits in a comfortable chair while the TMS coil is properly set in place on their head. (While this may sound intimidating, it really isn’t!) After the headpiece is in place, electromagnetic pulses are sent to the prefrontal cortex, which is the area of the brain responsible for mood.
While the magnetic pulses are being sent to the brain, patients can watch TV, listen to music, or talk with our staff. If they want and are comfortable, they can also have a friend or family member in the room with them during their TMS treatment session.
Each treatment lasts about 20-30 minutes. The full course of treatment will typically be five days a week for four to six weeks. Generally, patients will feel just like their normal selves after their treatment sessions. They will be able to drive and go back to their regular activities. Some people report feeling a little tired or having a mild headache, but that usually goes away after the first week or two. Other people report more energy and better focus, as well as the ability to ignore obsessive, anxious thoughts after treatment.
What Our Patients Are Saying about the Power of TMS Therapy
"TMS gave me a life back."
Christy, TMS Patient
Hear from a Mindful Health Solutions patient on how Transcranial Magnetic Stimulation improved her mental well-being and transformed her outlook on life.
Your Path to Transcranial Magnetic Stimulation Treatment
Is your antidepressant not working, you're experiencing an antidepressant failure, or you have bad medication side effects? In order to qualify for TMS treatments, you have to have tried at least two medication management plans and either not experienced relief of your symptoms or experienced uncomfortable side effects from them. If that is the case, your provider will talk to you about integrating TMS therapy into your customized treatment plan.
It is important to understand that mental health treatment plans are not a one-size-fits-all situation. Because of this situation, depression treatment plans for Transcranial Magnetic Stimulation may take longer or require different treatment option combinations to work best for certain patients. It is important for patients to communicate clearly with their provider about how their treatment plan is working. Collaboration between patient and provider can greatly impact the results of a patient’s wellness journey.





We're Paving the Way in Depression Care with Transcranial Magnetic Stimulation
Mindful Health Solutions is the nation’s leader provider of TMS therapy to patients like you. In fact, we founded one of the first TMS-focused clinics in the US and have deep expertise in the field of non-invasive brain stimulation. Patients at Mindful Health Solutions have access to true thought leaders in the field focused on bringing academic rigor and years of experience with novel therapeutics such as Transcranial Magnetic Stimulation to an integrated treatment strategy.
If you are considering TMS treatment, you can rest assured that you will get the most evidence-based, safe, and effective treatment possible with us at Mindful Health Solutions.

Why Choose Mindful Health Solutions for Transcranial Magnetic Stimulation Therapy?
Sadly, depression is an all-too-common condition that impacts millions of people in the United States alone. We want to bring that number down one person at a time, starting with you.
Overall, we understand the importance of patient-centered care and taking a true individualized approach. Patients respond well to the dynamic menu of treatments we offer, which includes traditional medication management as well as cutting-edge alternatives such as Transcranial Magnetic Stimulation (TMS) for depression, Spravato (Esketamine), and Ketamine IV Therapy.
Mindful Health Solutions also wants to make treatment comfortable and accessible. We offer telepsychiatry if patients are physically unable to come into the office or prefer a virtual appointment. This option allows patients to get care from wherever they are in the state.
Insurance
We are in-network with Kaiser, Medicare, and most major commercial insurance plans.
Frequently Asked Questions
What Is TMS?
TMS (Transcranial Magnetic Stimulation) is a non-invasive treatment for depression that uses gentle magnetic pulses to stimulate areas of the brain involved in mood regulation.
Is TMS safe?
Yes. TMS is FDA-cleared and has been used safely for many years in the treatment of depression.
How does TMS work?
In people with depression, certain brain regions may be underactive. TMS delivers targeted magnetic pulses to help activate and rebalance these areas, which can improve mood over time and help alleviate depression symptoms. TMS also encourages the brain to regulate activity on its own.
Still wondering, "How does TMS work?" Reach out today for a consultation!
Who is TMS for?
TMS is often recommended for people with depression who haven’t found enough relief from antidepressant medications or therapy alone. A clinical evaluation helps determine if it’s right for you.
Transcranial Magnetic Stimulation (TMS) for anxiety is available as well. This non-invasive brain stimulation treatment uses magnetic pulses to regulate overactive and underactive brain areas involved in mood, helping reduce anxiety symptoms by promoting healthier neural activity. The goal is to calm hyperactivity in fear-processing centers and strengthen emoptional regulation.
What does a TMS session feel like?
Most people describe it as a light tapping or clicking sensation on the scalp. Sessions are generally comfortable and well tolerated.
How long does each treatment take?
TMS is a clinic-based, outpatient procedure. No anesthesia is used, so patients can drive themselves to and from their appointments and resume their normal activities right away.
A typical session lasts about 20–30 minutes, and you’re awake the entire time. A typical course of treatment consists of 30 to 36 sessions administered over a 4-6 week time period.
How successful is TMS?
TMS introduces a new era of hope into our patients' lives. Most patients with treatment-resistant depression respond favorably to TMS, and many achieve full remission even after years of illness. Many patients maintain their results well over a year after treatment, though some may require periodic follow-up sessions. Medication and psychotherapy are often more effective after a course of TMS and may help maintain the long-term therapeutic benefit.
How long before I see results?
Some people begin to notice improvements within a few weeks, while others may take longer. Results build gradually over the course of treatment.
How is TMS better than medications?
While medications are an invaluable tool in treating depression, they don’t work for everyone. If you have tried more than one antidepressant with no relief, or have experienced uncomfortable side effects, TMS for depression is a proven option for you. TMS only targets certain areas of the brain that impact mood regulation, meaning it doesn’t cause side effects associated with medication. Most importantly, patients who failed two antidepressants are twice as likely to achieve remission with TMS therapy, meaning their symptoms completely go away.
What are the side effects of TMS?
TMS is not medication and doesn’t have systemic side effects like weight gain or sedation. The most common side effect is mild scalp discomfort during treatment, which usually improves quickly.
Some patients find the headpiece uncomfortable and may experience mild headaches during the first few treatments, but these symptoms typically subside within two weeks. Most side effects, such as headaches, fatigue, lightheadedness, scalp discomfort, and facial muscle twitching or tingling, are mild and short-lasting. Serious TMS side effects of repetitive treatments are rare, and our expert providers use their professional knowledge to minimize any risks.
Is TMS the same thing as ECT?
Not at all! While both TMS and Electroconvulsive Therapy (ECT) are brain stimulation treatments for depression, they work very differently:
- ECT uses an electrical current to induce a controlled seizure, requiring general anesthesia in a hospital. It’s typically used for severe, life-threatening depression and can cause memory loss.
- TMS uses magnetic fields to stimulate a specific brain area without inducing seizures or requiring anesthesia. It has minimal side effects, and patients can resume daily activities immediately.
Discover a new era of TMS with Mindful Health Solutions. TMS is a non-invasive outpatient option with fewer risks, making it a preferred choice for many individuals.
Do I need anesthesia or recovery time?
No. TMS does not require anesthesia, and you can drive yourself home and return to normal activities right after each session.
What does TMS treat?
Since its FDA approval as a depression treatment in 2008, further studies have shown that TMS has the potential to treat a wide range of conditions, many of which can be debilitating. Although the Mindful Health Solutions team focuses primarily on alleviating the symptoms of treatment-resistant depression, our clinics also provide TMS therapy for anxiety, autism in adults, bipolar disorder, body dysmorphia, certain types of chronic pain, memory disorders, migraines, mild cognitive impairment, mild dementia, multiple sclerosis, obsessive-compulsive disorder (OCD), Parkinson’s disease, PTSD, and smoking cessation. There is also evidence that TMS may help in recovery from stroke. Research and studies on the impact of TMS are continuing, and we at Mindful Health Solutions are excited to be at the forefront of this innovative therapy.
Is Transcranial Magnetic Stimulation covered by insurance?
Wondering, is tms covered by insurance?
Yes! Transcranial Magnetic Stimulation (TMS) is generally covered by most major insurance providers, including Blue Cross Blue Shield, Aetna, and UnitedHealthcare, for Trearment-Resistant Depression (TRD). Coverage and eligibility are reviewed during your consultation, and typically requires documented and unsuccessful attempts with traditional antidepressants and/or therapy.
